What Is CIP Cleaning and How Does It Work? The Ultimate 2026 Guide
Imagine shutting down your entire production line, dismantling heavy stainless steel piping, and sending a crew with scrub brushes into a confined, hazardous tank. Decades ago, this was the only way to ensure industrial hygiene. Today, that approach is not only a massive drain on profitability but also a severe safety liability. In modern Food & Beverage, Pharmaceutical, and Chemical manufacturing, operational downtime is the enemy, and cross-contamination is a brand-destroying catastrophe.
This brings us to a fundamental question for plant managers and process engineers: What exactly is CIP (Clean-in-Place), and how does it guarantee 100% sanitary conditions without taking your equipment apart?
In this comprehensive guide, we will break down the fundamental mechanics of CIP systems. You will learn the core principles that govern effective cleaning, step-by-step cycle breakdowns, how to eliminate dangerous "dead legs," and how selecting the right equipment can drastically reduce your water and chemical usage. Before diving into the technical mechanics, understanding why tank cleaning is critical is the foundational step toward optimizing your plant's operational efficiency, ensuring product safety, and complying with stringent FDA and GMP regulations.

Table of Contents
- 1. Understanding CIP Cleaning: The Basics
- 2. Core Concepts Simplified
- 3. Step-by-Step Guide: The Standard CIP Cycle
- 4. Expert Tips & Common Pitfalls to Avoid
- 5. Conclusion & Final Thoughts
1. Understanding CIP Cleaning: The Basics
Clean-in-Place (CIP) is an automated engineering methodology used to clean the interior surfaces of pipes, vessels, process equipment, filters, and associated fittings without requiring the disassembly of the system.
For facilities operating in English-speaking regions (US, UK, CA, AU) where labor costs are high and regulatory scrutiny is intense, CIP is not merely a convenience—it is a critical compliance tool. Regulatory bodies like the FDA (Food and Drug Administration) and guidelines under GMP (Good Manufacturing Practices) require scientifically validated proof that a processing environment is free from microbial contaminants and chemical residues between production batches.
Why CIP is the Industry Standard:
- Drastic Downtime Reduction: What used to take a full shift of manual labor can now be accomplished in 60 to 90 minutes.
- Uncompromising Consistency: Automated systems eliminate human error. Every cycle runs with the exact same temperature, chemical concentration, and duration.
- Sustainability and Cost Savings: Modern CIP systems are designed to recover and reuse water and chemicals, significantly lowering the environmental footprint and operational expenditure (OpEx).
- Operator Safety: Confined space entry is one of the most dangerous activities in an industrial plant. CIP keeps your workforce outside the tanks and away from harsh, high-temperature chemicals.
2. Core Concepts Simplified
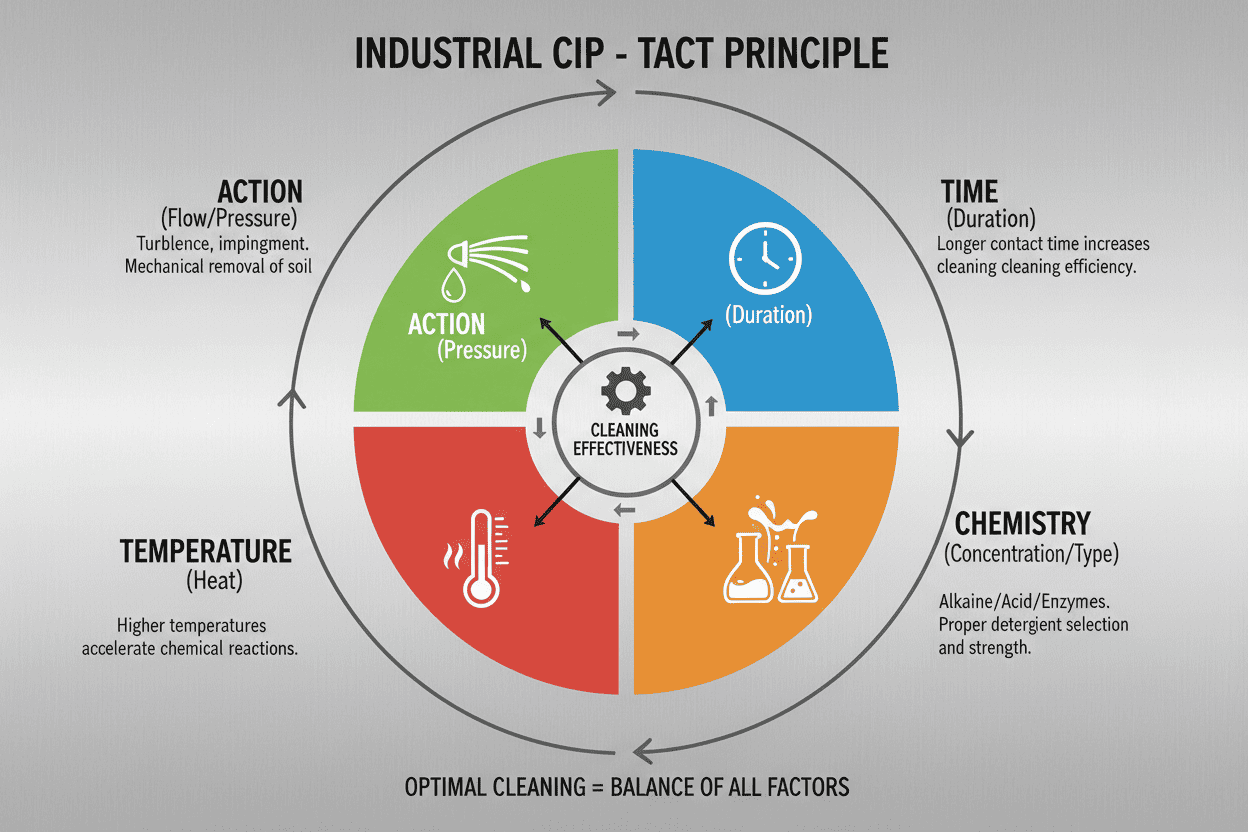
To truly grasp how CIP systems achieve microscopic cleanliness, we need to strip away the complex engineering jargon. The success of any automated cleaning system relies on a delicate balance of four elements, universally known as the TACT Principle.
Think of the TACT principle exactly like washing greasy dishes in your kitchen sink. If you use cold water instead of hot, you will have to scrub much harder or use a much stronger dish soap to get the plate clean. In industrial CIP, the same physics apply.
- T - Time: How long the cleaning solution is circulated against the soiled surfaces. Longer times can compensate for weaker chemicals.
- A - Action: The mechanical, physical force used to blast away sticky residues. In a tank, this "scrubbing" action is delivered by high-pressure spray nozzles.
- C - Chemistry: The specific alkaline (caustic) or acidic solutions used to dissolve proteins, fats, and mineral scales.
- T - Temperature: The heat level of the fluids. Higher temperatures exponentially increase the effectiveness of the chemicals and help melt viscous fats.
Demystifying Industry Jargon
Before upgrading a system, plant managers must understand the specific terminology used by process engineers:
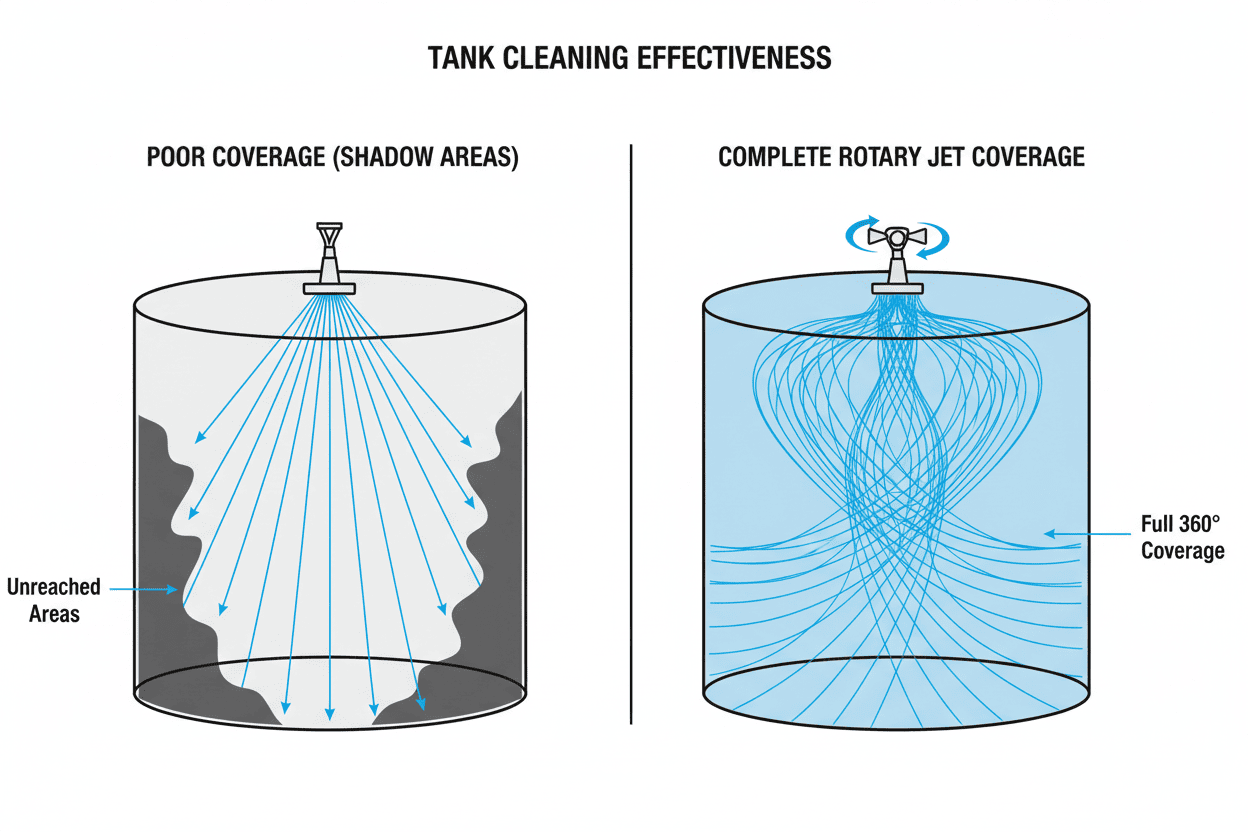
- Dead Legs / Shadow Areas: These are the hard-to-reach pockets inside pipes or tanks—such as behind agitator blades or inside complex valves. If the spray nozzle's water jet cannot physically hit these spots, bacteria will survive, grow, and contaminate the next batch.
- Impingement: This is the physical "punch" or impact force of the water jet hitting the stainless steel wall. High impingement is required to physically blast away tough, baked-on, or sticky residues (like yeast in breweries or thick syrups in food processing).
- CIP Validation: In highly regulated sectors like Pharmaceuticals, you cannot simply say a tank is clean. You must prove it. Validation is the rigorous, documented testing process (using swab tests and rinse water analysis) to scientifically guarantee that zero active pharmaceutical ingredients (APIs) or bacteria remain.
The Great Debate: Old vs. New
When facility managers analyze their operational bottlenecks, they frequently weigh the pros and cons of their current setup. A deep dive into manual vs automated tank cleaning reveals why heavily regulated industries have entirely abandoned hand-scrubbing.
Comparison Table: Manual vs. Automated CIP Systems
| Feature/Metric | Manual Tank Cleaning | Automated CIP System |
|---|---|---|
| Cleaning Consistency | Highly variable (depends on operator fatigue) | 100% repeatable and precise |
| Downtime / Labor Time | High (Requires disassembly and confined entry) | Low (Push-button operation, rapid turnaround) |
| Water & Chemical Waste | Extremely High (Continuous manual spraying) | Low (Systems recover and recirculate fluids) |
| Safety Risks | Critical (Confined space, chemical exposure) | Minimal (Fully enclosed operation) |
| Regulatory Validation | Nearly impossible to validate scientifically | Highly documentable, GMP/FDA compliant |
3. Step-by-Step Guide: The Standard CIP Cycle
To make the complex industrial process relatable, imagine your home washing machine. It doesn't just fill with soapy water once; it goes through a calculated series of fills, agitations, and rinses to ensure the clothes are clean and free of detergent. A standard industrial CIP cycle operates on the exact same logic, usually involving 5 to 7 distinct steps.
Phase 1: The Pre-Rinse
The cycle begins by flushing the system with water (often recycled from the final rinse of the previous cycle to save costs).
- Purpose: To mechanically blast away loose, bulk soils, un-dissolved sugars, and large particles. If warm water is used, it helps melt fats.
- Crucial Detail: Skipping this step means your expensive chemicals in the next phase will be wasted fighting loose dirt instead of breaking down bonded residues.
Phase 2: The Caustic Wash
This is the heavy-lifting phase. A highly alkaline solution (typically Sodium Hydroxide / Caustic Soda, ranging from 1% to 2% concentration) is circulated at high temperatures (often between 60°C to 85°C).
- Purpose: The caustic chemistry saponifies fats (turns them into soap) and breaks down complex protein structures, allowing them to be washed away.
Phase 3: Intermediate Rinse
Fresh or recovered water is pumped through the system to flush out the caustic chemicals and the suspended soils.
- Purpose: Prepares the tank for the acid wash. Mixing caustic and acid directly would neutralize both and create a dangerous chemical reaction.
Phase 4: The Acid Wash (Optional but Recommended)
Depending on the industry, an acid solution (like Nitric or Phosphoric acid) is circulated at lower temperatures.
- Purpose: While caustic removes organic material, acid removes inorganic material. It dissolves mineral scaling (like calcium deposits or "beer stone" in breweries) and neutralizes any lingering traces of the alkaline wash.
Phase 5: Final Rinse
The system is flushed with highly purified water—often Deionized (DI) or Reverse Osmosis (RO) water in pharmaceutical settings.
- Purpose: Ensures absolutely all chemical residues are flushed from the system. The rinse water is often tested for conductivity; if the conductivity matches pure water, the tank is chemically clean.
Phase 6: Sanitization / Sterilization
Before the next production batch begins, the system must be sterilized to kill any remaining microorganisms.
- Purpose: This is achieved either through chemical sanitizers (like Peracetic Acid) or by circulating live steam (a process known as SIP - Sterilization-in-Place).
Scenario A: Choosing the Right Equipment to Deliver the "Action"
You can have the perfect temperature, the strongest chemicals, and all the time in the world, but if the physical cleaning equipment inside the tank fails, the CIP cycle fails. The most critical component inside the processing vessel is the nozzle. It acts as the "showerhead," converting fluid pressure from the CIP pumps into the mechanical "Action" of the TACT principle.
Understanding how tank cleaning nozzles work is essential for any process engineer. If you use a weak nozzle in a large tank, the water will simply trickle down the walls, leaving sticky residues untouched.
For heavy-duty industrial applications dealing with viscous, sticky, or baked-on residues, static spray balls are no longer sufficient. Selecting high-impact tank cleaning nozzles for industrial applications is the most effective way to eliminate dead legs, guarantee CIP validation, and drastically reduce turnaround time.

Specifications Table: Choosing the Right Tank Cleaning Nozzle
| Nozzle Type | Mechanism of Action | Impingement (Impact Force) | Best Suited For | Water/Chemical Efficiency |
|---|---|---|---|---|
| Static Spray Balls | Cascading fluid down tank walls | Very Low | Light soils, easily soluble liquids (e.g., milk, juices) | Poor (Requires high volume of water) |
| Rotary Spray Heads | Spinning fan of fluid | Moderate | Medium soils, craft brewing, standard food processing | Good (Better coverage than static) |
| Rotary Jet Heads | 360° rotating high-pressure solid jets | Extremely High | Heavy, sticky, viscous, or baked-on residues (e.g., resins, peanut butter, APIs) | Excellent (Uses mechanical force over water volume) |
4. Expert Tips & Common Pitfalls to Avoid
If you spend time on industry forums like Reddit's r/ChemicalEngineering or r/TheBrewery, you will quickly realize that real-world CIP operations rarely go perfectly on the first try. Theory is one thing; practice is another. Here are the most common pitfalls plant managers face and how to avoid them.
Pitfall 1: The "Invisible" Shadow Areas (Dead Legs)
The Problem: A QA engineer fails a CIP validation swab test because bacteria were found behind the tank's agitator paddle. The standard spray ball simply couldn't spray behind the physical obstruction. The Fix: Never rely on a single spray ball for a complex tank. Use multiple nozzles strategically placed to create overlapping spray patterns, or upgrade to a 3D Rotary Jet Head that maps the entire interior volume over a set cycle time.
Pitfall 2: Clogged Static Spray Balls
The Problem: Operators notice the tank isn't getting clean. Upon inspection, they find that the tiny holes in the static spray ball are clogged with hop matter, fruit pulp, or debris from the process. The Fix: Implement an inline strainer before the CIP pump to catch debris. Better yet, replace static spray balls with self-cleaning rotary heads that do not have micro-perforations prone to clogging.
Pitfall 3: Ignoring Pump Pressure Drops
The Problem: The CIP system is designed to deliver 5 bar of pressure to the nozzle, but the actual impact inside the tank is weak. The Fix: Process engineers often forget to calculate friction loss in the piping between the pump and the tank. Always install pressure gauges at the nozzle inlet, not just at the pump discharge, to ensure the TACT "Action" parameter is actually being met.
5. Conclusion & Final Thoughts
Mastering your CIP cleaning process is one of the highest-ROI investments a manufacturing facility can make. It is the invisible backbone of modern industrial hygiene, protecting both your brand's reputation and your bottom line.
By deeply understanding the TACT principle, respecting the strict sequence of the CIP cycle, and migrating from outdated manual methods to fully automated solutions, you eliminate the guesswork from plant sanitation. Most importantly, recognizing that the mechanical force of your tank cleaning nozzles dictates the success of your entire multi-million-dollar CIP infrastructure will save you countless hours of troubleshooting and failed validations.
Quick Summary: CIP Optimization Checklist
| Focus Area | Key Takeaway | Immediate Action Step |
|---|---|---|
| Mechanics | CIP relies on TACT: Time, Action, Chemistry, Temperature. | Audit your current cycle: Are you relying too heavily on chemicals because your mechanical "Action" is weak? |
| Equipment | Manual cleaning is obsolete; automation ensures compliance. | Review labor costs and downtime associated with manual scrubbing to justify an automated CIP upgrade. |
| Nozzles | Static spray balls waste water; Rotary jets save resources. | Inspect your tanks for "shadow areas" and upgrade to high-impact rotary nozzles where necessary. |
| Validation | You must prove cleanliness for FDA/GMP compliance. | Ensure your final rinse water conductivity matches pure water baselines before releasing the tank for production. |
Ready to eliminate dead legs, reduce your water consumption, and optimize your cleaning cycles? Evaluate your current nozzle setup today and ensure your facility is operating at peak efficiency.






