The Difference Between SCR and SNCR Systems Explained: A Complete Guide for Plant Managers
For plant managers, industrial engineers, and environmental compliance officers, navigating the alphabet soup of emission control technologies is a high-stakes task. As the US EPA and EU directives tighten their grip on Nitrogen Oxide (NOx) emissions, facilities are forced to upgrade their infrastructure. The dilemma usually boils down to one critical question: What is the exact difference between SCR and SNCR systems, and which one will keep my facility compliant without destroying my CapEx budget?
Choosing the wrong system can lead to millions of dollars in wasted capital, exorbitant operational costs (OpEx), or crippling fines for failing to meet local environmental standards. In this comprehensive guide, we will break down the bottom-line differences, compare the ROI of both technologies, and explain how denitrification works in plain English. By the end of this article, you will have a clear, data-driven framework to decide which system is the right fit for your specific operational needs.

Table of Contents
- 1. Understanding SCR and SNCR Systems: The Basics
- 2. Core Concepts Simplified: Catalyst vs. High Heat
- 3. Step-by-Step Guide to Choosing Between SCR and SNCR
- 4. Expert Tips & Common Pitfalls to Avoid
- 5. Conclusion & Final Thoughts
1. Understanding SCR and SNCR Systems: The Basics
Before we dive into the technical comparison, we must understand the enemy: NOx (Nitrogen Oxides). These are toxic gases produced when fossil fuels (coal, oil, natural gas) or waste materials are burned at extremely high temperatures in boilers, kilns, or incinerators. NOx is the primary culprit behind urban smog and acid rain. Because of its severe environmental and health impacts, regulatory bodies worldwide strictly monitor and limit its release.
To combat NOx, industrial facilities utilize a process called denitrification, which involves injecting a chemical reagent (usually ammonia or urea) into the exhaust gas. This chemical reacts with the NOx, breaking it down into harmless nitrogen gas and water vapor before it exits the smokestack.
The two dominant technologies used to achieve this are SCR (Selective Catalytic Reduction) and SNCR (Selective Non-Catalytic Reduction). While both systems use the same basic chemical principles to neutralize NOx, how they trigger that reaction—and the price tag attached to that mechanism—are vastly different. For a procurement manager or plant engineer, understanding this distinction is the key to balancing environmental compliance with operational profitability.
2. Core Concepts Simplified: Catalyst vs. High Heat
To make the difference between SCR and SNCR perfectly clear, let’s use a vehicle analogy.
Think of an SCR system as a high-performance sports car. It is expensive to purchase upfront, requires precise engineering and specialized parts (the catalyst), but it delivers unmatched performance, achieving up to 95% NOx reduction. Conversely, think of an SNCR system as a reliable, heavy-duty pickup truck. It is much cheaper to buy and install, lacks complex internal components, and relies on "brute force" (extreme heat) rather than finesse. However, its performance ceiling is lower, typically achieving only a 30% to 70% reduction in NOx.
The Role of the Catalyst (The SCR Shortcut)
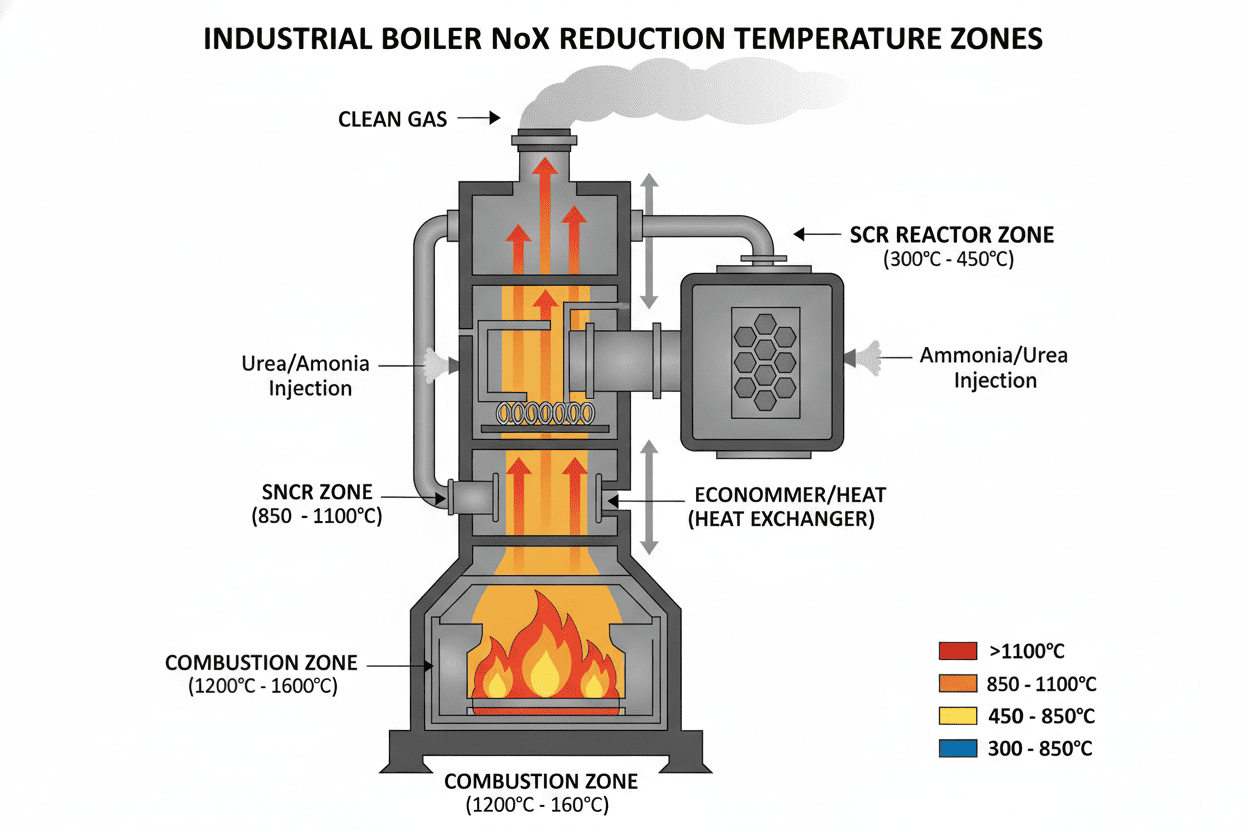
A Catalyst is a special material—often a honeycomb or corrugated structure made from metals like titanium, tungsten, or vanadium. In an SCR system, the exhaust gas and the injected reagent pass through this catalyst. The catalyst acts as a chemical shortcut, speeding up the reaction and allowing the ammonia to neutralize the NOx at much lower, more manageable exhaust temperatures (typically 500°F to 800°F).
The Goldilocks Zone of High Heat (The SNCR Method)
SNCR systems do not use a catalyst. Because they lack this chemical shortcut, they require a massive amount of thermal energy to force the chemical reaction to occur. SNCR systems must inject the reagent directly into the boiler or furnace where temperatures are extreme—specifically within a "Goldilocks zone" of 1600°F to 2100°F.
- If the temperature is too high, the ammonia actually burns and creates more NOx.
- If the temperature is too low, the reaction doesn't happen, leading to a dangerous and wasteful phenomenon known as "ammonia slip."
Core Concept Comparison Table
| Feature | SCR (Selective Catalytic Reduction) | SNCR (Selective Non-Catalytic Reduction) |
|---|---|---|
| Primary Mechanism | Uses a metallic catalyst to drive the reaction. | Uses extreme furnace heat to drive the reaction. |
| Operating Temperature | Low: 500°F - 800°F (260°C - 427°C) | High: 1600°F - 2100°F (870°C - 1150°C) |
| NOx Reduction Efficiency | High: 70% to 95%+ (EPA validated) | Moderate: 30% to 70% (EPA validated) |
| Space Requirement | Requires significant footprint for catalyst housing. | Minimal footprint; direct injection into boiler. |
| System Complexity | High (Requires soot blowers, catalyst management). | Low (Primarily pumps, piping, and nozzles). |
3. Step-by-Step Guide to Choosing Between SCR and SNCR
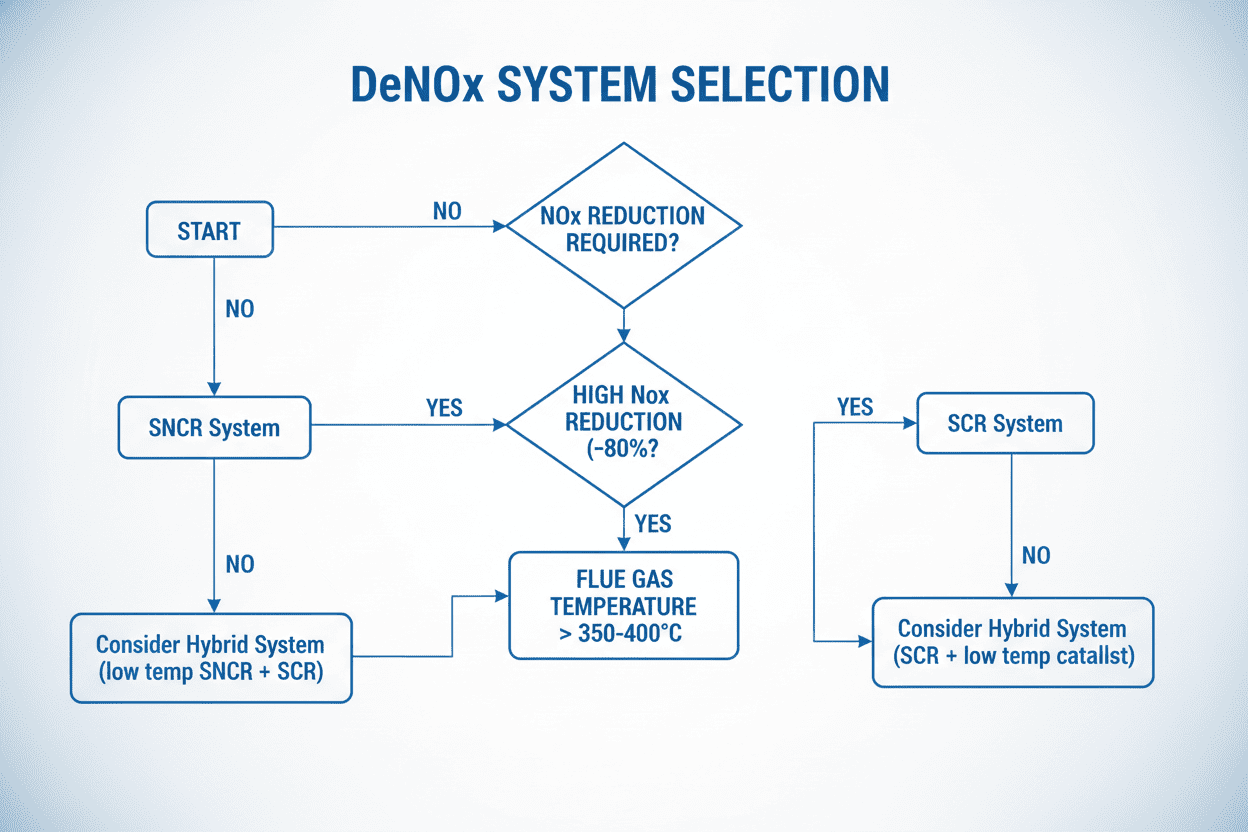
Selecting the right system requires a careful audit of your facility's baseline emissions, local regulatory targets, and budget constraints. Here is a practical framework to guide your decision-making process.
3.1 Scenario A: Strict Regulations & High NOx Reduction (The SCR Route)
If your facility is located in a highly regulated zone (e.g., California, strict EU zones) and you are required to slash NOx emissions by 80% or more, SCR is your only viable option. While the initial Capital Expenditure (CapEx) is daunting—often running into the millions due to the cost of the catalyst beds and structural modifications—the Operational Expenditure (OpEx) is highly efficient. Because the catalyst ensures almost all the injected ammonia reacts with the NOx, chemical waste is minimized.
Best for: Large power plants, heavy manufacturing facilities, and plants with stable, continuous operations.
3.2 Scenario B: Budget-Conscious Retrofits & Moderate Targets (The SNCR Route)
If your regulatory target only requires a 40% to 50% reduction in NOx, installing an SCR system would be massive financial overkill. SNCR is the perfect fit here. The CapEx for SNCR is a fraction of SCR because there is no catalyst to buy and no massive housing structure to build. You simply install injection lances directly into the upper furnace. However, plant managers must be aware that SNCR has a higher OpEx over time. Because the reaction is less efficient without a catalyst, you must inject significantly more reagent to achieve the desired reduction, leading to higher ongoing chemical costs.
Best for: Cement kilns, municipal solid waste incinerators, and smaller industrial boilers facing moderate regulatory updates.
3.3 The Hybrid Approach (SNCR/SCR)
For facilities that need high efficiency but lack the physical space for a massive full-scale SCR catalyst bed, a hybrid system is becoming increasingly popular. This involves installing an SNCR system in the furnace to knock down the bulk of the NOx (e.g., 40%), followed by a small, in-duct SCR catalyst downstream to "polish" the remaining exhaust and capture any unreacted ammonia.
Specification and Financial Data Table for System Selection
| System Type | CapEx (Initial Investment) | OpEx (Ongoing Costs) | Reagent Consumption | Maintenance Focus |
|---|---|---|---|---|
| SCR | $$$$ (Very High) | $$ (Moderate) | Highly Efficient (Low Waste) | Catalyst cleaning/replacement, draft fan power. |
| SNCR | $ (Low) | $$$ (High) | Less Efficient (High Volume) | Nozzle wear, boiler tube corrosion, chemical supply. |
| Hybrid | $$$ (Moderate/High) | $$ (Moderate) | Optimized | Balanced maintenance of both systems. |
4. Expert Tips & Common Pitfalls to Avoid
Even the most expensive, perfectly engineered emission control system will fail if operational execution is poor. Based on decades of industrial engineering experience and common troubleshooting threads found in plant management forums, here are the critical pitfalls you must avoid.
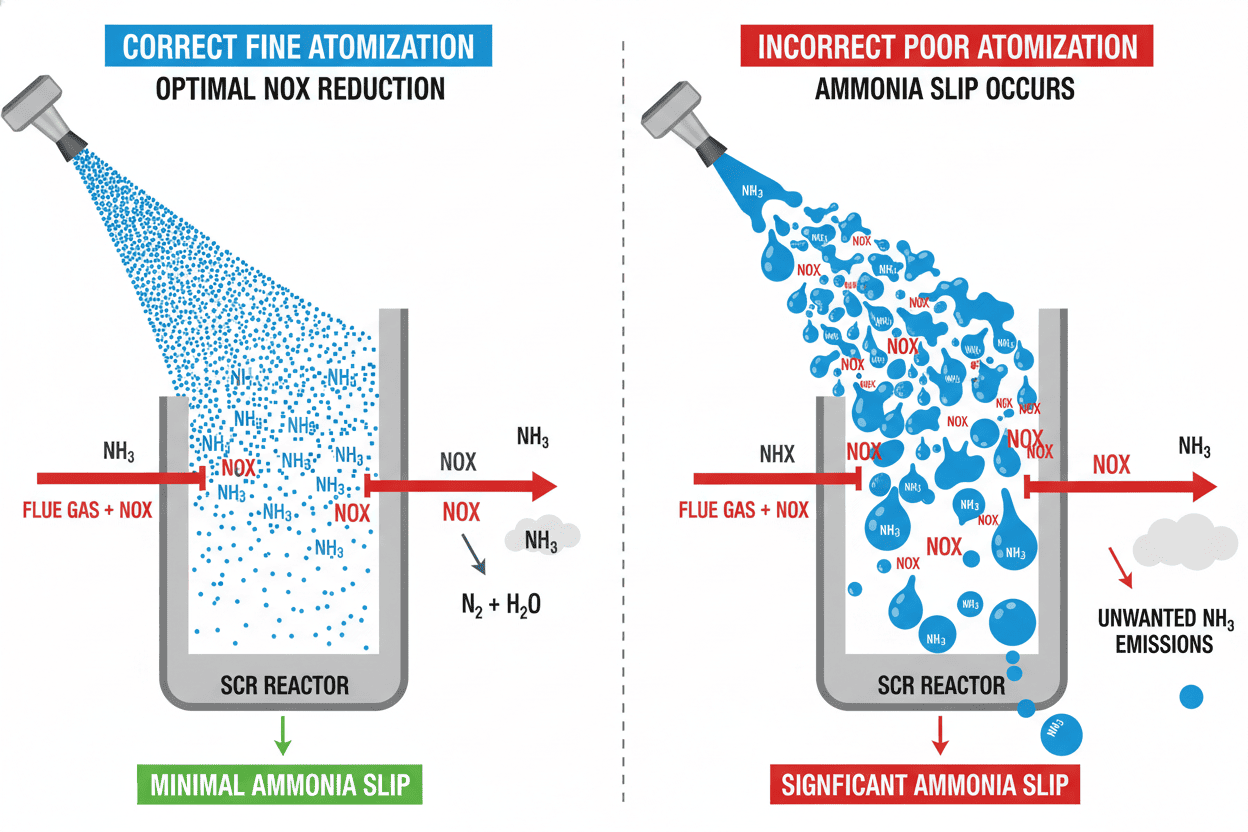
🛑 Pitfall 1: Ignoring the Critical Role of Injection Nozzles
This is the most common and costly mistake plant engineers make. The best SNCR or SCR system in the world will fail completely if the spray nozzles clog, degrade, or distribute the reagent unevenly.
In both systems, the ammonia or urea must be atomized into incredibly fine droplets.
- If the droplets are too large, they won't evaporate and react in time. They will crash into boiler tubes, causing severe corrosion and unexpected downtime.
- If the spray pattern is uneven, pockets of NOx will escape untreated, causing you to fail compliance tests.
To protect your investment, you must utilize high-efficiency SCR and SNCR denitrification nozzles designed specifically for emission control. These precision-engineered nozzles ensure optimal droplet size and uniform distribution, maximizing the chemical reaction and protecting your boiler infrastructure from liquid impingement.
🛑 Pitfall 2: Allowing "Ammonia Slip"
Ammonia Slip occurs when the injected reagent (ammonia or urea) fails to react with the NOx and simply escapes out of the smokestack into the atmosphere. Why is this a nightmare for plant managers?
- Wasted Money: You are literally spraying expensive chemicals into the sky for zero benefit.
- New Pollution: Ammonia is a pollutant itself. Emitting it can trigger secondary fines from environmental agencies.
- Equipment Damage: Unreacted ammonia can react with sulfur in the exhaust to form ammonium bisulfate, a sticky, highly corrosive substance that destroys downstream equipment like air preheaters.
Preventing ammonia slip requires strict temperature control (especially in SNCR) and, as mentioned above, flawless atomization through high-quality injection nozzles.
🛑 Pitfall 3: Treating NOx in Isolation
NOx is rarely the only pollutant in your exhaust gas. If your facility burns sulfur-heavy fuels (like certain grades of coal), you must also deal with Sulfur Dioxide (SO2). When designing an SCR or SNCR upgrade, engineers must consider how the system interacts with other emission controls, such as flue gas desulfurization (FGD) systems. Failing to integrate these systems holistically can lead to chemical cross-contamination and catastrophic equipment failure.
5. Conclusion & Final Thoughts
Deciding between an SCR and SNCR system is one of the most financially impactful choices a plant manager will make.
To summarize: If your regulatory environment demands maximum NOx reduction (70-95%+) and you have the capital budget to support it, SCR is the definitive choice. It operates at lower temperatures and offers excellent long-term reagent efficiency. However, if you are looking for a cost-effective retrofit to achieve moderate compliance (30-70%), SNCR is the practical, low-CapEx solution, provided you can manage the specific high-temperature injection requirements.
Regardless of which path you choose, remember that the success of your denitrification system hinges entirely on the quality of your reagent delivery. Investing in premium atomization technology is the cheapest insurance policy you can buy against ammonia slip, wasted chemicals, and regulatory fines.
Quick Summary Checklist
| Decision Factor | Choose SCR if... | Choose SNCR if... |
|---|---|---|
| Compliance Target | You need >70% NOx reduction. | You need <70% NOx reduction. |
| Budget Profile | High CapEx available; desire lower OpEx. | Tight CapEx budget; can absorb higher OpEx. |
| Space Constraints | You have space to build a catalyst housing. | Space is limited; need direct boiler injection. |
| Exhaust Temps | Gas is cooler (500°F - 800°F). | Furnace is extremely hot (1600°F - 2100°F). |
Next Steps: Are you currently evaluating an emission control upgrade or struggling with high ammonia slip in your existing setup? The problem likely lies at the point of injection. Evaluate your current nozzle performance and consult with engineering specialists to ensure your reagent delivery is optimized for your specific operational temperatures.






